Efficiency of Purified Biosurfactant (Pseudofactin) from P. Aeruginosa in Disruption of Biofilm Formation by Candida Albicans in Vaginal Candidiasis
Abstract
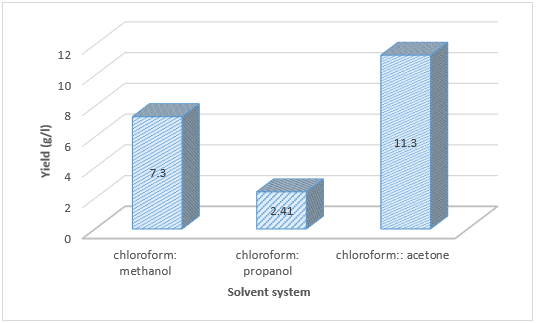
Candidiasis caused by Candida albicans is one of the most common forms of hospital-acquired opportunistic infection. So that the biosurfactant was detected in P. aeruginosa isolated from burns and wounds infections and produced with different levels of productivity. The use of (2:1) of chloroform: acetone was the best for extraction of biosurfactant with 11.3 g/l while chloroform: propanol gave a low yield of 2.41 g/l of biosurfactant. Candida albicans causing vaginal candidiasis considered a stronger producer of biofilms. The effect of biosurfactant on Candida biofilm formation was inhibited with increasing incubation period and the percentage of biofilm inhibition reached 45-78% after 24 hours and increased to 65-84% after 48 hours.
References
Nami, S., Mohammadi, R., Vakili, M., Khezripour, K., Mirzael, H. and Morovati, H. (2018). Fungal vaccines, mechanism of actions and immunology: a comprehensive review. Biomed Pharm.; 109:333– 344.
Sakagami, T., Kawano, T., Yamashita, K., Ymada, E., Fujino, N., Kaeriyama, M., Fukuda, Y., Nomura, N. (2019). Antifungal susceptibility trend and analysis of resistance mechanism for Candida species isolated from bloodstream at a Japanese university hospital. J Infect Chemother 25:34– 40.
Armstrong-James, D., Brown, G.D., Netea, M.G., Zelante, T., Gresnigt, M.S., van de Veerdonk, F.L. and Levitz, S.M. (2017). Immunotherapeutic approaches to treatment of fungal diseases. Lancet Infect Dis.; 17:e393– e402.
Shields, R.K., Kline, E.G., Healey, K.R., Kordalewska, M., Perlin, D.S., Nguyen, M.H. and Clancya, C.J. (2019). Spontaneous mutational frequency and FKS mutation rates vary by echinocandin agent against Candida glabrata. Antimicrob Agents Chemother 63:1– 9.
Ghannoum, M.A., Roilides, E., Katragkou, A., Petraitis, V. and Walsh, T.J. (2015). The role of echinocandins in Candida biofilm-related vascular catheter infections: In vitro and in vivo model systems. Clin Infect Dis.; 61:S618– S621.
Rajendran, R., Sherry, L., Nile, C.J., Sherriff, A., Johnson, E.M., Hanson, M.F., Williams, C., Munro, C.A. (2016). Biofilm formation is a risk factor for mortality in patients with Candida albicans bloodstream infection-Scotland, 2012–2013. Clin. Microbiol. Infect.; 22:87– 93.
Satpute, S.K.; Kulkarni, G.R. and Banpurkar, A.G.(2016). Biosurfactant from Lactobacilli species: Properties, challenges and potential biomedical applications. J Basic Microbiol.; 56 (11): 1140-1158.
Rodrigues, L.; Moldes, A.; Teixeria, J.; Oliveira, R.(2006). Kinetic study of fermentative biosurfactant production by Lactobacillus strains. Biochem Eng J.; 28:109-116.
Sharma, D.; Singh, S.B.; Shailly, K. (2016). Biosurfactants of lactic acid bacteria. ISBN. 978-3-319-26215-4. Springer Briefs in Microbiology. (Springer International Publishing), Switzerland;17-29.
Cornea, C.P.; Israel, R.; Sicuia, O.A.; Voaides, C.; Zamfir, M. and Simona, G(2014). Biosurfactant production by Lactobacillus spp. Strains isolated from Romanian traditional fermented food products. Rom Biotechnol Lett.;21(2):11312-11320.
Lin, J.; Chen, Y.; Xu, R. and Jia, Y. (2013).Screening and Evaluation of Biosurfactant-Producing Strains Isolated from Oilfield Wastewater. Indian J. Microbiol.; 53(2): 168–174.
Abouseoud, M.; Maachi, R.; Amrane, A.; Boudergua, S. and Nabi, A. (2008). Evaluation of different carbon and nitrogen sources in production of biosurfactant by Pseudomonas fluorescence. J. Desalination.; 223: 143-151.
Ibrahim, A.H. and Mahmood, M.S.(2020). Elicitation of Biosurfactant Production of Serratia Marcessens by Using Biotic and Abiotic Factors. Sys. Rev. Pharm.;11(11):1630-1638
Marinho, S.A.; Teixeira, A.B.; Santos, O.S. and Oliveira, S.D.(2010). Identification of Candida spp. by phenotypic tests and PCR. Braz. J. Microbiol.; 41(2):1517-8382.
Marak, M.B. and Dhanashree, B.(2018). Antifungal Susceptibility and Biofilm Production of Candida spp. Isolated from Clinical Samples. International Journal of Microbiology, 5 pages
Ryan, K.J. and Ray, C.G.(2004). Medical microbiology. McGraw Hill, 4: 370.
Deziel, E.; Paquette, G.; Villemur, R.; Lepine, F. and Bisaillon, J.(1996). Biosurfactant Production by a Soil Pseudomonas Strain Growing on Polycyclic Aromatic Hydrocarbons. Appli. Environ. Microbiol. 62(6):1908–1912.
Ron, E.Z. and Rosenberg, E.(2002).Biosurfactants and oil bioremediation. Curr. Opin. Biotechnol.,13(3):249-52.
Cho, S.K.; Shim,S.H.; Park, K.R.; Chio, S. and Lee.(2006). Purification and characterization of a biosurfactant produced by Pseudomonas sp. G11 by a symmetrical flow field fractionation(AsFIFFF).Analytical and Bioanalytical Chemistry, 386:2027-2033.
Nicholls, S., MacCallum, D.M., Kaffarnik, F.A., Selway, L., Peck, S.C. and Brown, A.J. (2011) Activation of the heat shock transcription factor Hsf1 is essential for the full virulence of the fungal pathogen Candida albicans. Fungal Genet Biol 48:297– 305.
Haque, F.; Alfatah, M.; Ganesan, K. and Bhattacharyya, M.S.(2016). Inhibitory Effect of Sophorolipid on Candida albicans Biofilm Formation and Hyphal Growth. Sci. Reports, 6: 23-28.